TechInvention partners with Eubiologics to launch India’s first oral cholera vaccine in LDPE
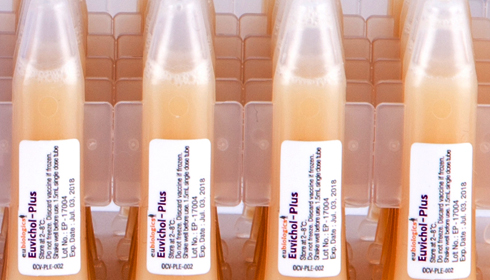
Mumbai-based startup TechInvention Lifecare has recently announced that it has recently launched Euvichol-Plus, the world's first and only oral cholera vaccine (OCV), to India in collaboration with South Korea based Eubiologics in a low-density polyethene (LDPE) single dose pack.
Announcing the launch, the company informed that the LDPE single dose pack is not only easy and safe to use, but it also reduces the issues pertaining to conventional glass vials in terms of breakage, storage, transportation, distribution, waste management, and facilitates delivery in emergencies and humanitarian crises in campaign mode.
The availability of the lifesaving vaccine that could save thousands of lives has been jeopardised at a time when more countries are reporting outbreaks due to the discontinuation of production by one of the world's only two manufacturers of the vaccine.
According to data from the World Health Organisation, the mortality associated with cholera outbreaks is of particular concern as many countries reported higher case fatality rates (CFR) than in previous years. The average cholera CFR, reported globally in 2021 was 1.9% (2.9% in Africa), a significant increase above acceptable (<1%) and the highest recorded in over a decade.
India features at the top of the list of cholera-endemic countries, with an estimated incidence rate of 1.64 per 1,000 people. In India, estimates show that 400 million people are at risk of cholera, with an estimated 6,751,888 cases and 20,256 (3%) deaths annually.
Also Read:
| - Doctors stress on clean water for drinking to prevent diseases |
From 2011 to 2020, 565 outbreaks were reported in India, which led to 45,759 cases and 263 deaths. This represents only the tip of the iceberg, as cholera remains an under-recognised health issue and is grossly under-reported in India. An effective cholera vaccine has long been the vision for controlling cholera in India.
“In this joint effort with Eubiologics to bring a preventive solution to the recurring endemic that is cholera, the launch of Euvichol-Plus is the empirical choice," Founder and CEO of TechInvention, Mr Syed Ahmed said.
“We wish to contribute to the national initiatives along the lines of WHO's ‘Ending Cholera: A Global Roadmap by 2030’,” he added while stating that the company has two more vaccines in late-stage clinical development and regulatory approval in India.
Commenting on the collaboration, Eubiologics' CEO, Mr Yeong-Ok Baik, stated, “We are happy to have collaborated with TechInvention and to bring our WHO-prequalified cholera vaccine, Euvichol-Plus, in a unidose pack to India.”
“We hope to make a difference in the lives of Indian children and contribute to the reduction of disease prevalence by increasing vaccine coverage wherever it is needed. The positive outcome of the clinical study encourages us to take part in similar initiatives,” he added.
Responding to a query by Drug Today Medical Times Mr Khan said that though India tops the list of cholera-endemic countries, it is not supported by GAVI, The Vaccine Alliance for oral cholera vaccine (OCV).
“Hence, it is the onus of the public procurement systems and private practitioners to facilitate vaccination,” he added.
Pointing out that Euvichol-Plus is the world’s first oral cholera vaccine (OCV) supplied in a plastic tube, it reduces the volume by nearly 30% and halves the weight Mr Khan said that the vaccine offers easier storage, transportation, wastage management, and facilitates delivery in emergencies or humanitarian campaigns.
“This makes administration much easier and the Phase 3 trial shows Euvichol-Plus is immunogenic, safe, and non-inferior to Sanofi’s cholera vaccine in healthy Indian adults and children,” he added.
The vaccine can be a game changer, in the sense that it will be highly effective in combatting the cholera outbreak settings, especially in conflict or natural disaster situations,” Mr Khan said.